1. 不(bu)銹鋼化學鍍銅的應用
不(bu)銹(xiu)鋼化學鍍銅(tong)應用(yong)于(yu)電子工業、計算機工業及航空工業中電子元件的高效電磁干擾的屏蔽。
2. 不銹鋼基體上化(hua)學(xue)鍍銅存在的問題
香蕉app免費視頻在線觀看:不銹鋼基體上化學鍍銅易造成鍍層鼓泡,這不僅影響了鍍層與基體的結合力,而且直接影響到外觀質量。為此,將鍍前酸處理過的不銹鋼放在烘箱中加熱,以除去酸洗時滲入到基體的氫,采用此方法解決了鍍層起泡問題,得到所需要的化學鍍銅層。
3. 不銹鋼化學鍍銅(tong)工藝(yi)流程
NiCr不銹鋼(gang)(經過600℃真空熱(re)處(chu)(chu)理)→化學除油[氫氧化鈉(na)(NaOH)10%(質量分數)]→水洗(xi)(xi)→熱(re)水洗(xi)(xi)→除銹(鹽酸(suan)1:1溶液,溫度(du)80~100℃,時間(jian)5min)→水洗(xi)(xi)→干燥(zao)→除氫(在烘箱中溫度(du)200℃,時間(jian)2h)→酸(suan)處(chu)(chu)理[稀硫酸(suan)5%(質量分數),時間(jian)1~5min]→水洗(xi)(xi)→去離子(zi)水洗(xi)(xi)→化學鍍銅(tong)→水洗(xi)(xi)→抗銅(tong)變色處(chu)(chu)理(苯并三氮唑1g/L,溫度(du)65℃,時間(jian)2min)→純水洗(xi)(xi)→熱(re)純水洗(xi)(xi)→干燥(zao)。
4. 化(hua)學鍍(du)銅溶液成分及工藝條件見表4-39

5. 化學(xue)鍍(du)銅溶(rong)液(ye)的配(pei)制
先將(jiang)(jiang)硫酸(suan)銅和(he)酒(jiu)(jiu)石酸(suan)鉀(jia)鈉分別用(yong)純(chun)水溶(rong)(rong)(rong)(rong)解(jie)(jie),然后將(jiang)(jiang)硫酸(suan)銅溶(rong)(rong)(rong)(rong)液(ye)(ye)在(zai)攪(jiao)拌下(xia)(xia)加(jia)(jia)入酒(jiu)(jiu)石酸(suan)鉀(jia)鈉溶(rong)(rong)(rong)(rong)液(ye)(ye)中,銅離子被酒(jiu)(jiu)石酸(suan)離子絡(luo)(luo)合(he)成藍(lan)色絡(luo)(luo)合(he)物。再將(jiang)(jiang)氯化鎳用(yong)少量水溶(rong)(rong)(rong)(rong)解(jie)(jie)后攪(jiao)拌加(jia)(jia)入,再加(jia)(jia)入甲醛溶(rong)(rong)(rong)(rong)液(ye)(ye),攪(jiao)拌均勻(yun)。將(jiang)(jiang)氫(qing)氧化鈉用(yong)純(chun)水溶(rong)(rong)(rong)(rong)解(jie)(jie)成200g/L 的濃溶(rong)(rong)(rong)(rong)液(ye)(ye)待用(yong)。在(zai)開(kai)始化學鍍銅前,逐步(bu)在(zai)攪(jiao)拌下(xia)(xia)加(jia)(jia)入藍(lan)色絡(luo)(luo)合(he)液(ye)(ye)中,使溶(rong)(rong)(rong)(rong)液(ye)(ye)pH達到(dao)12 左右(用(yong)9~13精密pH試紙測量),最后將(jiang)(jiang)穩定劑亞鐵氯化鉀(jia)、聚(ju)乙二醇(chun)用(yong)少量水溶(rong)(rong)(rong)(rong)解(jie)(jie)后攪(jiao)拌加(jia)(jia)入,乙醇(chun)可(ke)直接(jie)加(jia)(jia)入,最后用(yong)純(chun)水加(jia)(jia)入至(zhi)溶(rong)(rong)(rong)(rong)液(ye)(ye)的規定體(ti)積(ji),攪(jiao)拌均勻(yun)后放入不銹(xiu)鋼件即(ji)可(ke)開(kai)始化學鍍銅。
6. 操作(zuo)要(yao)點
①. 裝載量
按照每升鍍液裝載2d㎡計算。
②. 除氫和攪(jiao)拌
不銹鋼(gang)(gang)對氫(qing)(qing)(qing)滲很(hen)敏感,工件在(zai)(zai)酸洗過(guo)(guo)程中氫(qing)(qing)(qing)會(hui)滲人到基(ji)體中,如果不除氫(qing)(qing)(qing),化(hua)學(xue)(xue)鍍(du)銅(tong)(tong)鍍(du)層(ceng)致密小孔覆蓋在(zai)(zai)不銹鋼(gang)(gang)表面后(hou),氫(qing)(qing)(qing)氣(qi)無(wu)法(fa)逸(yi)出(chu),造成(cheng)(cheng)很(hen)大的(de)(de)應(ying)力,使鍍(du)層(ceng)起泡(pao),加上化(hua)學(xue)(xue)鍍(du)銅(tong)(tong)本身伴隨著析氫(qing)(qing)(qing)過(guo)(guo)程,氫(qing)(qing)(qing)氣(qi)會(hui)殘留在(zai)(zai)基(ji)體與鍍(du)層(ceng)金屬的(de)(de)晶格中,增大內(nei)應(ying)力,嚴重地減弱基(ji)體與鍍(du)層(ceng)的(de)(de)結合(he)強度(du)(du)。為(wei)此,從兩方(fang)面著手解(jie)決鍍(du)層(ceng)起泡(pao)問(wen)題。其一(yi)是把經過(guo)(guo)去油(you)、酸洗后(hou)的(de)(de)工件在(zai)(zai)化(hua)學(xue)(xue)鍍(du)銅(tong)(tong)前(qian)進行熱處理(li)(li),除去滲入到基(ji)體中的(de)(de)氫(qing)(qing)(qing),熱處理(li)(li)溫(wen)(wen)度(du)(du)和(he)時(shi)間條件經實驗確(que)定(ding)(ding)為(wei)180~200℃,2小時(shi),鍍(du)層(ceng)無(wu)鼓泡(pao),鍍(du)層(ceng)結合(he)力合(he)格。溫(wen)(wen)度(du)(du)過(guo)(guo)低或時(shi)間過(guo)(guo)短仍有(you)輕微(wei)鼓泡(pao),溫(wen)(wen)度(du)(du)過(guo)(guo)高或時(shi)間過(guo)(guo)長都容易(yi)使表面再次生(sheng)成(cheng)(cheng)不易(yi)去除的(de)(de)氧化(hua)皮,又需要較(jiao)長時(shi)間的(de)(de)強酸處理(li)(li),酸洗時(shi)氫(qing)(qing)(qing)會(hui)再次滲入基(ji)體。在(zai)(zai)所選(xuan)定(ding)(ding)的(de)(de)溫(wen)(wen)度(du)(du)和(he)時(shi)間下(xia)雖表面會(hui)有(you)新的(de)(de)氧化(hua)膜(mo)生(sheng)成(cheng)(cheng),但使用(yong)稀硫酸短時(shi)間酸洗即可,以免(mian)再次滲氫(qing)(qing)(qing)。其二是在(zai)(zai)化(hua)學(xue)(xue)鍍(du)銅(tong)(tong)過(guo)(guo)程中,采用(yong)某種攪(jiao)拌(空(kong)氣(qi)攪(jiao)拌或機械攪(jiao)拌),有(you)利(li)于銅(tong)(tong)離子向工件表面擴散,防止和(he)減少副反應(ying)產(chan)物銅(tong)(tong)粉(即Cu2O)的(de)(de)生(sheng)成(cheng)(cheng),而(er)且有(you)利(li)于反應(ying)產(chan)物氫(qing)(qing)(qing)氣(qi)脫離工件表面。通過(guo)(guo)上述兩種方(fang)法(fa)有(you)效(xiao)地解(jie)決了鍍(du)層(ceng)鼓泡(pao)問(wen)題,提高了鍍(du)層(ceng)與基(ji)體的(de)(de)結合(he)強度(du)(du)。
③. 催化活性劑-鎳離子
在(zai)化(hua)學鍍(du)銅溶液中加入少量(liang)鎳離子(zi)后,鍍(du)層(ceng)性(xing)質得到(dao)改善,在(zai)鍍(du)銅層(ceng)中含有微(wei)量(liang)的鎳,形成Cu89Ni11金屬化(hua)合物,它具(ju)有最佳的催化(hua)活性(xing),提高鍍(du)層(ceng)的催化(hua)活性(xing)。
④. 穩定劑(ji)的(de)控制
在化(hua)(hua)學鍍(du)(du)(du)銅(tong)(tong)過(guo)程中(zhong)(zhong),甲(jia)(jia)醛能將二價銅(tong)(tong)離子還原為(wei)金(jin)屬銅(tong)(tong)鍍(du)(du)(du)層,還存在有(you)副反(fan)應(ying),即不完(wan)全反(fan)應(ying)生(sheng)成(cheng)暗紅(hong)色的(de)(de)(de)氧(yang)化(hua)(hua)亞銅(tong)(tong)(Cu2O),它(ta)形成(cheng)微(wei)粒懸浮在鍍(du)(du)(du)液(ye)中(zhong)(zhong),呈膠體狀態,極難用(yong)過(guo)濾除(chu)去,若與(yu)銅(tong)(tong)共沉積,使銅(tong)(tong)鍍(du)(du)(du)層疏松粗糙,與(yu)基體結合力極差。氧(yang)化(hua)(hua)亞銅(tong)(tong)被(bei)甲(jia)(jia)醛還原成(cheng)金(jin)屬微(wei)粒,又(you)成(cheng)為(wei)自催(cui)化(hua)(hua)中(zhong)(zhong)心,使鍍(du)(du)(du)液(ye)自發(fa)分(fen)(fen)解(jie),消耗了鍍(du)(du)(du)液(ye)中(zhong)(zhong)的(de)(de)(de)有(you)效成(cheng)分(fen)(fen)。為(wei)了抑制副反(fan)應(ying)的(de)(de)(de)發(fa)生(sheng),加入(ru)穩(wen)定(ding)(ding)劑,以提高鍍(du)(du)(du)液(ye)的(de)(de)(de)穩(wen)定(ding)(ding)性(xing)(xing)。但是,過(guo)量(liang)(liang)的(de)(de)(de)穩(wen)定(ding)(ding)劑的(de)(de)(de)加人,又(you)成(cheng)了化(hua)(hua)學鍍(du)(du)(du)銅(tong)(tong)反(fan)應(ying)的(de)(de)(de)催(cui)化(hua)(hua)毒性(xing)(xing)劑,顯著降低(di)化(hua)(hua)學鍍(du)(du)(du)的(de)(de)(de)速率(lv),甚至停鍍(du)(du)(du),故選用(yong)穩(wen)定(ding)(ding)劑,并控制其很低(di)的(de)(de)(de)適宜含量(liang)(liang),對提高鍍(du)(du)(du)液(ye)穩(wen)定(ding)(ding)性(xing)(xing)有(you)效。
⑤. 防銅(tong)層變色處理
對銅(tong)層(ceng)進行防變色(se)(se)處(chu)理,在鍍(du)銅(tong)層(ceng)表面形(xing)成(cheng)一層(ceng)穩定的(de)絡(luo)合膜,隔絕外界浸蝕性物(wu)質對鍍(du)銅(tong)層(ceng)的(de)作用,使鍍(du)銅(tong)層(ceng)保持(chi)本色(se)(se)一定的(de)時間。苯并三氮唑要(yao)先用乙醇溶解好,然后加(jia)入(ru)熱(re)蒸餾水中(zhong)。防變色(se)(se)處(chu)理的(de)溫度(du)不低于65℃,時間不少于2min,否則防變色(se)(se)達不到效果(guo)。
7. 鍍層結合強度(du)檢測-劃痕實驗法
在鍍層(ceng)表(biao)面(mian)用刀(dao)片劃出1mm間距的直(zhi)行線(xian)和90°交錯的橫行線(xian)形成小方格。觀察劃痕交錯處鍍層(ceng)有(you)無起層(ceng),進一步用黏(nian)性高(gao)的膠(jiao)帶(dai)貼于劃痕表(biao)面(mian),再撕下膠(jiao)帶(dai),以(yi)銅層(ceng)不脫落為合(he)格。
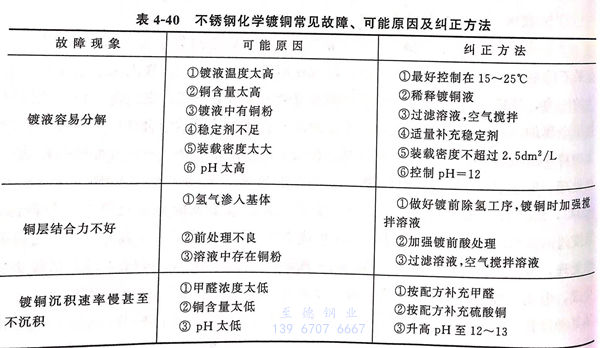
8. 不銹鋼化學鍍銅常見故障、可能原因及糾(jiu)正方法見表(biao)4-40.