不同熱處理溫度下鐵素體單一相在0.5mol/L 硫酸溶液中的極化曲線如圖5.62所示,從圖中可以看出,鐵素體單一相在0.5mol/L 硫酸溶液中的極化曲線形貌與2205雙相(xiang)不銹鋼不同,雙相(xiang)不銹鋼整體在0.5mol/L 硫酸溶液中的極化曲線陽極區具有明顯的活化鈍化區以及很寬電位范圍的鈍化區,而鐵素體單一相沒有活化鈍化區,且鈍化區比較窄。具體擬合數據如表5.20所列,腐蝕電位和自腐蝕電流隨固溶溫度的變化曲線如圖5.63所示。
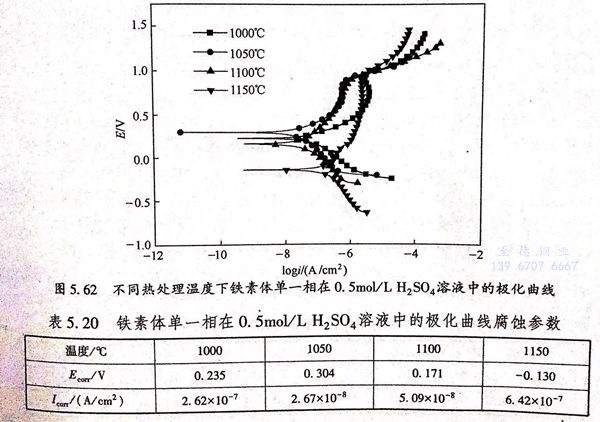
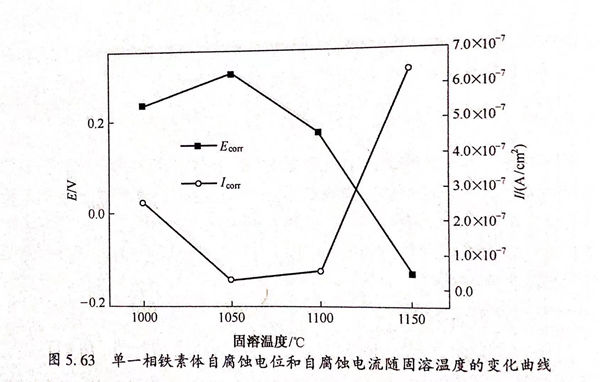
結合圖5.63和表5.20分析可知,鐵素體單一相的自腐蝕電位隨固溶溫度的增大,后減小,當固溶溫度為1050℃時,Ecorr的值達到最大為0.304V;當固溶溫度達到1150℃后,Bm的值達到最小為-0.130V.自腐蝕電流的變化規律同自腐蝕電位相反,為先減小,后增大,于1050℃達到最小值2.67×10-8A/c㎡,于1150℃達到最大值6.42×10-7A/c㎡.結合自腐蝕電位和自腐蝕電流的變化趨勢可知,當固溶溫度為1050℃時,鐵素體單一相在0.5mol/L 硫酸溶液中耐蝕性能最佳,隨著固溶溫度的升高,其耐蝕性變差。這與雙相不銹鋼整體在0.5mol/L 硫酸溶液中的耐蝕規律相一致。產生以上現象的主要原因1050℃固溶后,鐵素體Cr元素和Mo元素含量最高,Cr元素是鈍化膜形成的要元素,并且可以提高鈍化膜的致密度和修復速度。有研究表明,金屬表面化膜的性質與Cr/Fe的值和金屬/氧的值有關,Cr/Fe、金屬/氧的比值越高,膜的耐蝕性能越好,而Cr元素含量提高會使Cr/Fe、金屬/氧比升高。Mo元素可以配合Cr元素提高材料的耐蝕性能。隨著溫度的升高,鐵素體含量的升高以及Cr和Mo含量的降低導致平均單位中Cr和Mo含量的降低,相當于Cr和Mo被不斷稀釋,因此,當固溶溫度達到1150℃后,鐵素體單一相的耐蝕性變差。
圖(tu)5.64為(wei)不同固(gu)溶(rong)溫(wen)(wen)度下(xia)2205 雙(shuang)相(xiang)不銹鋼奧氏體(ti)單一(yi)相(xiang)在(zai)0.5mol/L 硫酸溶(rong)液中(zhong)的(de)極化(hua)(hua)曲(qu)(qu)線(xian)(xian),奧氏體(ti)單一(yi)相(xiang)極化(hua)(hua)曲(qu)(qu)線(xian)(xian)形(xing)狀與鐵素體(ti)單一(yi)相(xiang)相(xiang)似,均沒有(you)雙(shuang)相(xiang)不銹鋼整(zheng)體(ti)在(zai)0.5mml/1.1,50,溶(rong)液中(zhong)出現(xian)的(de)法化(hua)(hua)鈍化(hua)(hua)區以及(ji)很寬的(de)純化(hua)(hua)區間。當固(gu)溶(rong)溫(wen)(wen)度較低(di)時,剪氏體(ti)單一(yi)相(xiang)極化(hua)(hua)曲(qu)(qu)線(xian)(xian)靠近圖(tu)的(de)左上方,隨(sui)著(zhu)固(gu)溶(rong)溫(wen)(wen)度的(de)升高,曲(qu)(qu)線(xian)(xian)向右(you)下(xia)方移動,其具(ju)體(ti)批合數據如表(biao)5.20折(zhe)列,自腐蝕電位和自腐蝕電流(liu)隨(sui)溫(wen)(wen)度的(de)變化(hua)(hua)曲(qu)(qu)線(xian)(xian)如圖(tu)5.65所(suo)示(shi)。
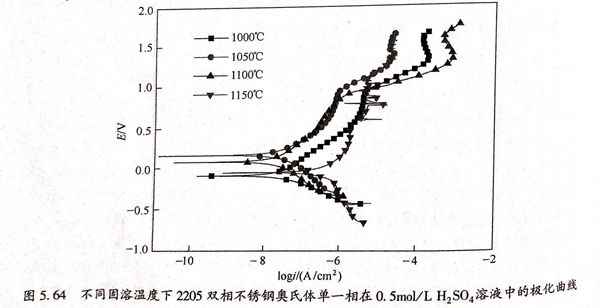
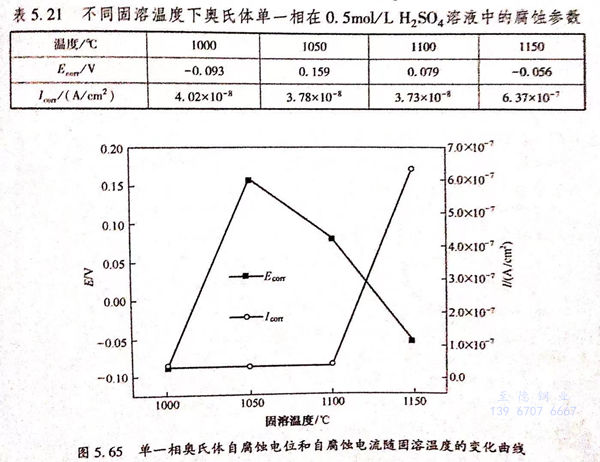
結合(he)表5.21和(he)圖5.65可(ke)知(zhi),奧氏(shi)(shi)體單一相(xiang)自(zi)腐(fu)蝕(shi)電位隨固溶(rong)(rong)溫(wen)(wen)度(du)(du)的升(sheng)高呈(cheng)現先增大后減小的趨勢,當(dang)固溶(rong)(rong)溫(wen)(wen)度(du)(du)為(wei)(wei)1050℃時,具有(you)最大值0.159V,隨著固溶(rong)(rong)溫(wen)(wen)度(du)(du)的升(sheng)高,自(zi)腐(fu)蝕(shi)電位變(bian)為(wei)(wei)1100℃的0.079V和(he)1150℃時的-0.056V,說明1050℃奧氏(shi)(shi)體單一相(xiang)在0.5mol/L 硫酸溶液中耐蝕傾向最好,隨著溫度的升高,耐蝕傾向變差。自腐蝕電流在固溶溫度較低時比較小,分別為1000℃的4.02×10-8A/c㎡,1050℃的3.78×10-8A/c㎡,1100℃的3.73×10-8A/c㎡,其值相差不多,當固溶溫度達到1150℃時,自腐蝕電流增加了一個數量級,其值為6.37×10~7A/c㎡.綜合自腐蝕電位和自腐蝕電流的變化趨勢可知,當固溶溫度較低時,奧氏體單一相在0.5mol/L硫酸溶液中的耐蝕性較好,當固溶溫度升高至1150℃后,其耐蝕性能變差。奧氏體單一相在0.5mol/L 硫酸溶(rong)液(ye)中(zhong)耐蝕(shi)性變化(hua)趨(qu)勢與在3.5%NaCl溶(rong)液(ye)中(zhong)相(xiang)同。當固溶(rong)溫度升高時,Cr、Mo元素在奧氏體中(zhong)的(de)含量變化(hua)不大,但是Ni元素含量的(de)下降,導致(zhi)奧氏體單一相(xiang)耐蝕(shi)性變差。
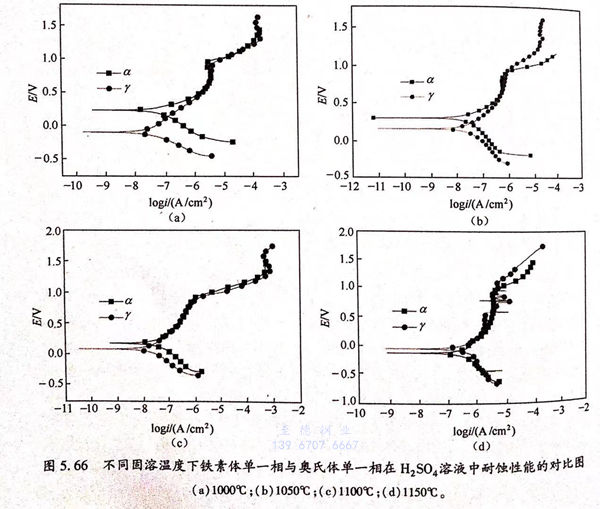
圖5.66為不(bu)同(tong)固(gu)溶(rong)(rong)(rong)(rong)溫(wen)度(du)下素體單(dan)(dan)一(yi)相(xiang)(xiang)與奧(ao)氏(shi)體單(dan)(dan)一(yi)相(xiang)(xiang)在(zai)(zai)(zai)硫(liu)酸(suan)溶(rong)(rong)(rong)(rong)液中(zhong)耐蝕(shi)(shi)性能的對比圖,從(cong)圖中(zhong)可以看出,固(gu)溶(rong)(rong)(rong)(rong)溫(wen)度(du)較(jiao)低(di)時,鐵(tie)(tie)素體單(dan)(dan)一(yi)相(xiang)(xiang)自(zi)(zi)腐蝕(shi)(shi)電(dian)(dian)位較(jiao)高(gao)。隨著溫(wen)度(du)的升高(gao),兩相(xiang)(xiang)自(zi)(zi)腐蝕(shi)(shi)電(dian)(dian)位差(cha)值變小,當固(gu)溶(rong)(rong)(rong)(rong)溫(wen)度(du)為1150℃時,奧(ao)氏(shi)體單(dan)(dan)一(yi)相(xiang)(xiang)自(zi)(zi)腐蝕(shi)(shi)電(dian)(dian)位較(jiao)鐵(tie)(tie)素體單(dan)(dan)一(yi)相(xiang)(xiang)略微提高(gao)。比較(jiao)不(bu)同(tong)固(gu)溶(rong)(rong)(rong)(rong)溫(wen)度(du)下的兩相(xiang)(xiang)的自(zi)(zi)腐蝕(shi)(shi)電(dian)(dian)流可以發現(xian)(xian),奧(ao)氏(shi)體單(dan)(dan)一(yi)相(xiang)(xiang)與鐵(tie)(tie)素體單(dan)(dan)一(yi)相(xiang)(xiang)的自(zi)(zi)腐蝕(shi)(shi)電(dian)(dian)流在(zai)(zai)(zai)不(bu)同(tong)固(gu)溶(rong)(rong)(rong)(rong)溫(wen)度(du)下都相(xiang)(xiang)差(cha)很(hen)小。相(xiang)(xiang)對在(zai)(zai)(zai)3.5%NaCl溶(rong)(rong)(rong)(rong)液,鐵(tie)(tie)素體單(dan)(dan)一(yi)相(xiang)(xiang)在(zai)(zai)(zai)0.5mol 硫(liu)酸(suan)溶(rong)(rong)(rong)(rong)液中(zhong)的腐蝕(shi)(shi)速率(lv)有(you)了很(hen)明顯的增(zeng)加,這(zhe)說明在(zai)(zai)(zai)0.5mol 硫(liu)酸(suan)溶(rong)(rong)(rong)(rong)液中(zhong),鐵(tie)(tie)素體單(dan)(dan)一(yi)相(xiang)(xiang)與奧(ao)氏(shi)體單(dan)(dan)一(yi)相(xiang)(xiang)都表現(xian)(xian)出了較(jiao)大的腐蝕(shi)(shi)活性,在(zai)(zai)(zai)宏觀上表現(xian)(xian)出均勻(yun)腐蝕(shi)(shi)的性質,其在(zai)(zai)(zai)0.5mol/L 硫(liu)酸(suan)溶(rong)(rong)(rong)(rong)液中(zhong)的優先腐蝕(shi)(shi)行為不(bu)明顯,這(zhe)與其在(zai)(zai)(zai)3.5%NaCl溶(rong)(rong)(rong)(rong)液中(zhong)的優先腐蝕(shi)(shi)行為明顯不(bu)同(tong)。